Fully-Automated
Semi-Automated
Radiology
Point of Care
Home » Global Programs » HPV Program » Cy-Tb
New test for latent Tb with confidence. Be it at hospital, clinic, or camps at grassroot or a national health program, latent Tb detection is not a challenge anymore.
Cy-Tb overcomes the problem of false positive in the previously BCG vaccinated Individuals
Unlike IGRA, no need for sample collection, transportation and testing in a controlled lab environment
Administered using widely popular Mantoux technique
• HIV-infected individual • Paediatric population
Performed in the field without any lab facility
Multidose vial is excellent for programmatic implementation
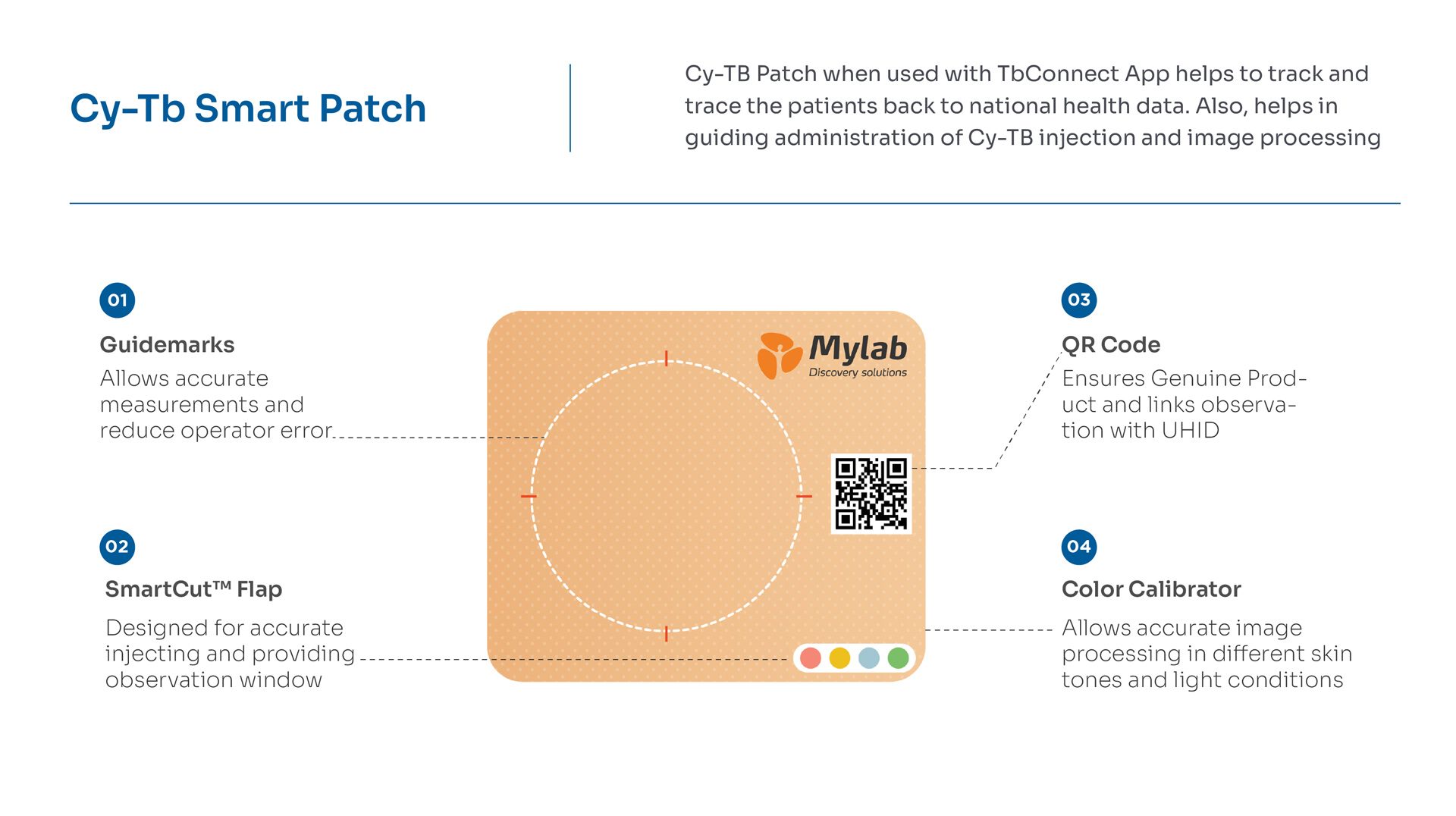
Patented smart patch to help track and trace the patients along with administration guidance
End-to-end App, from patient registration to interpretation of results in a consistent manner

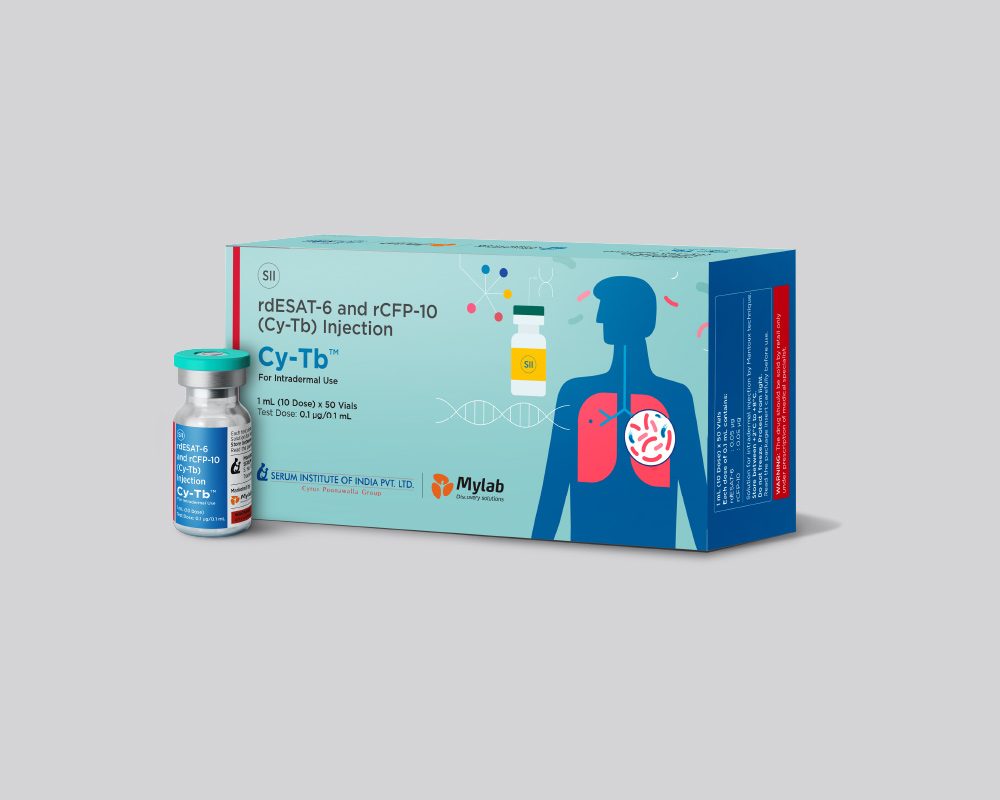
One dose (0.1 mL) contains:
• rdESAT-6: 50 ng
• rCFP-10: 50 ng
Clear, colourless to pale
yellow solution for injection

Diagnosis of latent TB infection with MTB in individuals of 18 years and above

The dosage is 0.1 mL

Intradermal injection using mantoux technique

Induration of ≥ 5 mm indicates latent infection

Store at 2-8°C
Shelf life of 18 months

No special training required

5th Floor, Amar Paradigm, Survey No – 110/11/3, Baner Rd, Pune – 411045. Maharashtra, India.